Morphed ViP3.x: Covering Population Variability
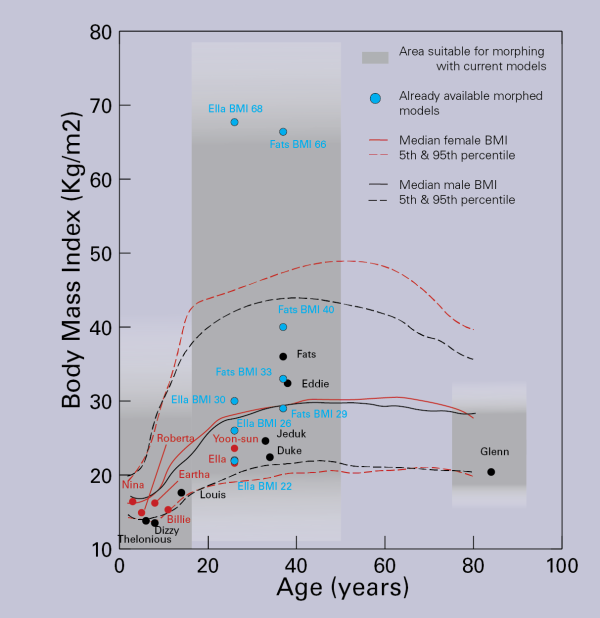
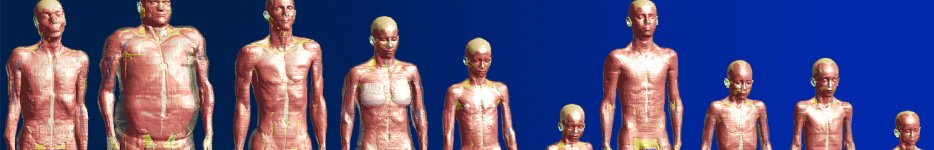
The morphed Virtual Population V3-x (mViP 3.1) models represent adapted versions of our existing models “Ella” and “Fats”. The adapted models include a selection of important gross anatomical descriptors, which allow investigations based on differences in e.g., the body mass index (BMI) or weight. Different morphed versions of Ella (22, 26, 30, 63 kg/m²) and Fats (29, 66, 70 kg/m²) are currently available. Additional versions can be created on demand.
Physics-based morphing allows to modify the volume of specific tissues (e.g., subcutaneous adipose tissue, muscle) based on a realistic biomechanics simulation. Volume change of a tissue is introduced using a thermal expansion analogy, leading to elastic deformation of surrounding tissues, constrained by rigid bones.
Breast-Coil MRI
For simulation applications related to breast coil design or safety, an additional modified model of Ella (Ella Breast Coil) is provided. The model incorporates a new high resolution segmentation of breast tissues segmented from an MRI breast scan. The breast shape is typical of a patient during a breast scan in prone position (Ella was in supine position). Additionally, the model's head/neck has been made posable.
| Name | Sex
|
Age |
Height1 |
Weight1 |
BMI1 |
|
female |
26 |
1.63 |
57.3 |
22 |
|
|
female |
26 |
1.63 |
69.4 |
26 |
|
|
female |
26 |
1.63 |
79.7 |
30 |
|
|
female |
26 |
1.63 |
167 |
63 |
|
|
female |
26 |
1.63 |
58.2 |
21.9 |
|
|
male |
37 |
1.82 |
96 |
29 |
|
|
male |
37 |
1.82 |
219 |
66 |
|
|
male |
37 |
1.82 |
232 |
70 |
1 These values are calculated based on the latest version of the models and of the tissue properties database
Systems Requirements
The above mentioned models require a 64-bit Windows XP and above operating system and at least 6 GB, respectively.
Distribution
Please address all inquiries regarding distribution to the Sim4Life sales team. To download the models, please click on any of the models in the table above to access the corresponding webpage. The download procedure will begin with a click on the model's DOI number.
Acknowledgements
The original ViP models were developed in cooperation with the Center for Devices and Radiological Health of the U.S. Food and Drug Administration, Silver Spring, MD, USA (FDA); the Austrian Research Centers GmbH, Seibersdorf, Austria; the University of Houston, TX, USA; the Hospital of the Friedrich-Alexander-University (FAU), Erlangen, Germany; and Siemens Medical Solutions, Erlangen, Germany.
The Mobile Manufacturers Forum, the GSM Association and SPEAG funded the development of two adult and two child models. The German Federal Office for Radiation Protection (BfS) supported the development of four additional child models. Scientists at FDA (USA) validated the models.
Contributions from The Netherlands Organization for Health Research and Development (ZonMw) supported the further development and significant improvement of the anatomical fidelity of the child models. Pediatricians at the Erasmus MC-Daniel den Hoed Cancer Center reviewed the models. The Swiss National Research Programme NRP 57 “Non-Ionising Radiation – Health and Environment” funded the development of the pregnant models.
References for citation
The Virtual Family – Development of Surface-based Anatomical Models of Two Adults and Two Children for Dosimetric Simulations
Andreas Christ, Wolfgang Kainz, Eckhart G. Hahn, Katharina Honegger, Marcel Zefferer, Esra Neufeld, Wolfgang Rascher, Rolf Janka, Werner Bautz, Ji Chen, Berthold Kiefer, Peter Schmitt, Hans-Peter Hollenbach, Jianxiang Shen, Michael Oberle, Dominik Szczerba, Anthony Kam, Joshua W. Guag and Niels Kuster, in Physics in Medicine and Biology, Volume 55, Issue 2, pp. N23–N38, January 2010
Development of a New Generation of High-Resolution Anatomical Models for Medical Device Evaluation: The Virtual Population 3.0
Marie-Christine Gosselin, Esra Neufeld, Heidi Moser, Eveline Huber, Silvia Farcito, Livia Gerber, Maria Jedensjo, Isabel Hilber, Fabienne Di Gennaro, Bryn Lloyd, Emilio Cherubini, Dominik Szczerba, Wolfgang Kainz and Niels Kuster, in Physics in Medicine and Biology, Volume 59, Issue 18, pp. 5287–5303, June 2014